Google Doc
Enzyme Lab
Testing Catalase Reactions
Laboratory 4, AP Biology
Abstract. We conducted labs over the course of two days to test catalase reactions and how a piece of chicken liver will interact with hydrogen peroxide. We put the chicken liver in multiple test tubes with different temperatures in each one. The temperatures being tested were hot (88 degrees Celcius), cold (3 degrees Celsius) and room temperature (24 degrees Celsius). We found that the heated test tube significantly sped up the reaction while the cold and room temperature test tubes slowed the process down.
Introduction
These labs were conducted to test catalase reactions and how chicken liver and hydrogen peroxide performed together. The first day was testing the reaction between chicken liver and hydrogen peroxide but the second day tested how the two reacted in different temperature settings. We also tested whether or not catalase is reusable. We initially hypothesized that catalase was not reusable, but at the end, catalase proved to be reusable. Through each test we discovered there was a fast reaction between the liver and the hydrogen peroxide. When we put the test tubes into different temperature settings we discovered temperature does make a difference. We came to the conclusion that heat sped the process up and that cold either halted or slowed the process down.
Methods
In the first day of the lab, we established our control group by: filling a test tube with 2 ml of hydrogen peroxide, cutting a relatively small piece of chicken liver, placing it down the test tube, then watching the reaction. Proceeding that we then separated the resulting solution and the sliver of chicken liver and placed them into separate test tubes. The two separate test tubes tested for two very different and important things. The test tube with the resulting solution of the first test had a new piece of chicken liver to it; this test was to prove what was residing within the reactant solution, and if it would react with new added catalyse. The second test, was to see if catalase was reuseable. We then added 2 more ml of hydrogen peroxide to the already used chicken liver to see if infact catalase would still produce the same initial reaction it did before. The second day we mimicked the procedures of the first day, but this time to prove what outside factors also contribute to the rate of of which cataclysm happens. We filled 3 test tubes with 2 ml of hydrogen peroxide, the first test tube would be left in room temperature (our control), the second would be placed in boiling water, and the third would be placed in a beaker filled with ice. We made sure to record the temperature of each setting: room temperature being in 24 degrees celsius, boiling water and 88 degrees celsius, and the beaker with ice at 3 degrees celsius. From there we proceeded to add a small piece of chicken liver to each of the different heat settings.
Results
When placing the liver in the test tube at room temperature the reaction begins almost immediately. On a scale of 1-5, the speed of the reaction takes place at a 4. We were able to determine these results by viewing the test tube as well as taking physical evidence with cameras. The contents of the test tube begin to bubble immediately and oxygen is released. This causes the hydrogen peroxide the remaining liquid to be simply water and the remaining residual catalase and hydrogen peroxide. The reaction was exothermic as heat was released and the test tube became slightly warmer. After placing more hydrogen peroxide it was observed that catalase, if still present, is reusable but hydrogen peroxide is not.
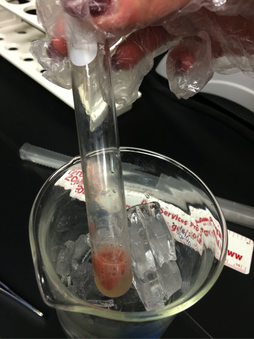
We conducted the experiment a second time but this time we added the variables of temperature. After placing the test tube in a boiling hot water bath at 88 degrees Celsius we once again added liver to the test tube. The reaction seemed to have speeded up to about a 5. Additionally, we placed the test tube in a beaker of ice at 3 degrees Celsius, we added liver to the test tube. We observed that the reaction had slowed down slightly placing the reaction rate at about a 3. Our results depict that heat accelerates the reaction whereas cold slows down the reaction.
Discussion
Enzymes are substances within the body that are responsible for catalysis. The lab helped us to better understand and observe first-hand the function of enzymes in nature. It also allowed us to see how enzymes functions in different conditions. We learned that enzymes break down hydrogen peroxide by the enzyme catalase in the textbook but were not able to gain practical applications of the chemical. The experiment allowed us to see the effect of temperature in the reaction, using both ice and boiling water as variables. Catalase helps to stop the reaction caused by the hydrogen peroxide in order to prevent damage to the substance.
The lab gave us the information that heat accelerates the catalase reaction. In order to corroborate our results we recommend that others conduct the experiment. In future experiments, it may be beneficial to observe the effects of different variables on the catalase reaction other than temperature.
Conclusion
These labs have effectively been able to prove that catalase reactions take place sooner when exposed to heat. The reactions are significantly slowed down or completely halted when exposed to a relatively cold temperature. Before we began the labs, we hypothesized that the catalase was not reusable. However, over the course of two labs, we were able to determine that catalase is in fact reusable. The results were different from what we had hypothesized.
Works Cited
Reece, C. (2014). AP Biology. Pearson.
Wootton, K. (2014). Enzyme Lab Notes. Coppell: Wootton, Kim.
Photos and Charts:
Enzyme Lab
Testing Catalase Reactions
Laboratory 4, AP Biology
Abstract. We conducted labs over the course of two days to test catalase reactions and how a piece of chicken liver will interact with hydrogen peroxide. We put the chicken liver in multiple test tubes with different temperatures in each one. The temperatures being tested were hot (88 degrees Celcius), cold (3 degrees Celsius) and room temperature (24 degrees Celsius). We found that the heated test tube significantly sped up the reaction while the cold and room temperature test tubes slowed the process down.
Introduction
These labs were conducted to test catalase reactions and how chicken liver and hydrogen peroxide performed together. The first day was testing the reaction between chicken liver and hydrogen peroxide but the second day tested how the two reacted in different temperature settings. We also tested whether or not catalase is reusable. We initially hypothesized that catalase was not reusable, but at the end, catalase proved to be reusable. Through each test we discovered there was a fast reaction between the liver and the hydrogen peroxide. When we put the test tubes into different temperature settings we discovered temperature does make a difference. We came to the conclusion that heat sped the process up and that cold either halted or slowed the process down.
Methods
In the first day of the lab, we established our control group by: filling a test tube with 2 ml of hydrogen peroxide, cutting a relatively small piece of chicken liver, placing it down the test tube, then watching the reaction. Proceeding that we then separated the resulting solution and the sliver of chicken liver and placed them into separate test tubes. The two separate test tubes tested for two very different and important things. The test tube with the resulting solution of the first test had a new piece of chicken liver to it; this test was to prove what was residing within the reactant solution, and if it would react with new added catalyse. The second test, was to see if catalase was reuseable. We then added 2 more ml of hydrogen peroxide to the already used chicken liver to see if infact catalase would still produce the same initial reaction it did before. The second day we mimicked the procedures of the first day, but this time to prove what outside factors also contribute to the rate of of which cataclysm happens. We filled 3 test tubes with 2 ml of hydrogen peroxide, the first test tube would be left in room temperature (our control), the second would be placed in boiling water, and the third would be placed in a beaker filled with ice. We made sure to record the temperature of each setting: room temperature being in 24 degrees celsius, boiling water and 88 degrees celsius, and the beaker with ice at 3 degrees celsius. From there we proceeded to add a small piece of chicken liver to each of the different heat settings.
Results
When placing the liver in the test tube at room temperature the reaction begins almost immediately. On a scale of 1-5, the speed of the reaction takes place at a 4. We were able to determine these results by viewing the test tube as well as taking physical evidence with cameras. The contents of the test tube begin to bubble immediately and oxygen is released. This causes the hydrogen peroxide the remaining liquid to be simply water and the remaining residual catalase and hydrogen peroxide. The reaction was exothermic as heat was released and the test tube became slightly warmer. After placing more hydrogen peroxide it was observed that catalase, if still present, is reusable but hydrogen peroxide is not.
We conducted the experiment a second time but this time we added the variables of temperature. After placing the test tube in a boiling hot water bath at 88 degrees Celsius we once again added liver to the test tube. The reaction seemed to have speeded up to about a 5. Additionally, we placed the test tube in a beaker of ice at 3 degrees Celsius, we added liver to the test tube. We observed that the reaction had slowed down slightly placing the reaction rate at about a 3. Our results depict that heat accelerates the reaction whereas cold slows down the reaction.
Discussion
Enzymes are substances within the body that are responsible for catalysis. The lab helped us to better understand and observe first-hand the function of enzymes in nature. It also allowed us to see how enzymes functions in different conditions. We learned that enzymes break down hydrogen peroxide by the enzyme catalase in the textbook but were not able to gain practical applications of the chemical. The experiment allowed us to see the effect of temperature in the reaction, using both ice and boiling water as variables. Catalase helps to stop the reaction caused by the hydrogen peroxide in order to prevent damage to the substance.
The lab gave us the information that heat accelerates the catalase reaction. In order to corroborate our results we recommend that others conduct the experiment. In future experiments, it may be beneficial to observe the effects of different variables on the catalase reaction other than temperature.
Conclusion
These labs have effectively been able to prove that catalase reactions take place sooner when exposed to heat. The reactions are significantly slowed down or completely halted when exposed to a relatively cold temperature. Before we began the labs, we hypothesized that the catalase was not reusable. However, over the course of two labs, we were able to determine that catalase is in fact reusable. The results were different from what we had hypothesized.
Works Cited
Reece, C. (2014). AP Biology. Pearson.
Wootton, K. (2014). Enzyme Lab Notes. Coppell: Wootton, Kim.
Photos and Charts: